![Rh(III)-catalyzed [5+1] oxidative cycloaddition of arylguanidines with alkynes a novel access to C4-disubstituted 1,4-dihydroquinazolin-2-amine](https://cdmf.org.br/wp-content/uploads/2019/04/RhIII-catalyzed-51-oxidative-cycloaddition-of-arylguanidines-with-alkynes-a-novel-access-to-C4-disubstituted-14-dihydroquinazolin-2-amine-518x381.png)
Rh(III)-catalyzed [5+1] oxidative cycloaddition of arylguanidines with alkynes: a novel access to C4-disubstituted 1,4-dihydroquinazolin-2-amines
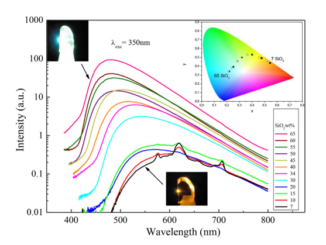
Abstract: Anovel and mild Rh-III-catalyzed [5+1] oxidative cycloaddition between arylguanidines and alkynes efficiently affords C4-disubstituted 1,4-dihydroquinazolin-2-amines. Members of this family of heterocycles, which contain the relevant cyclic guanidine units, have shown interesting pharmacological properties. The mechanism probably involves the formation of an eight-membered rhodacycle in which the imine unit of guanidine is coordinated to the Rh center. This rhodacycle would evolve to give the C-4 disubstituted 1,4-dihydroquinazolin-2-amine skeleton.
Author(s): Cajaraville, A; Suarez, J; Lopez, S; Varela, JA; Saa, C
CHEMICAL COMMUNICATIONS
Volume: 51 Pages: 15157-15160 Published: 2015
DOI: 10.1039/c5cc06388d