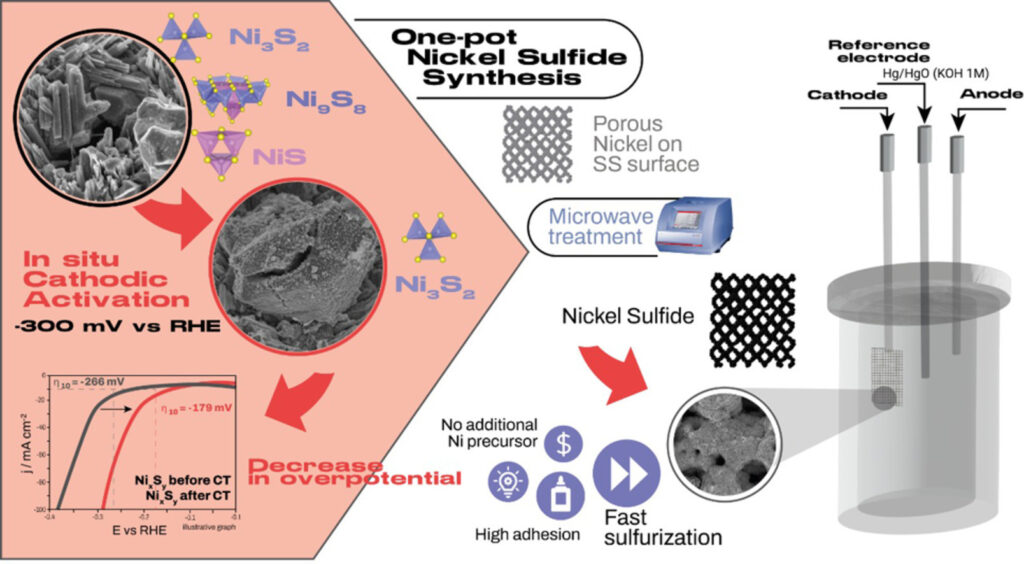
Abstract: Advancing alkaline water electrolysis requires bifunctional catalysts that are both low-cost and scalable. Here, we report a self-adaptive porous nickel sulfide (NixSy) electrode that, under cathodic operation, reconstructs in situ into a Ni3S2-rich phase via sulfur-vacancy formation. Porous Ni was first electrodeposited on stainless-steel mesh using a dynamic hydrogen-bubble template and then converted by a short, microwave-assisted sulfurization into a multiphase Ni-S precursor. Cathodic activation at -0.300 V vs RHE for 12 h in KOH selectively removes sulfur and stabilizes the Ni3S2 phase, enhancing conductivity and charge-transfer kinetics. The resulting vacancy-mediated phase evolution increases the electrochemically active surface area (ECSA) and yields superior catalytic behavior: overpotentials of -0.179 V (HER) and +0.251 V (OER) at ±10 mA cm⁻², with excellent stability over 100 h of continuous operation. In overall water electrolysis, the cell requires only 1.61 V at 10 mA cm⁻² and 1.89 V at 100 mA cm⁻², demonstrating bifunctional efficiency. Through rapid microwave synthesis and phase control, we produce durable Ni-S catalysts and directly connect their performance to the structural evolution guided by sulfur vacancies.
Graphical Abstract:
Author(s): Mitchell G.S. Da Silva, Matheus M. Brito, Gleison N. Marques, Lucia H. Mascaro
First published: 20/01/2026