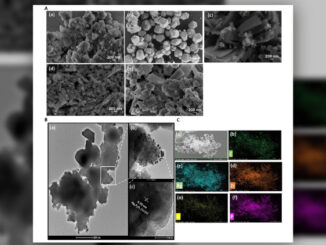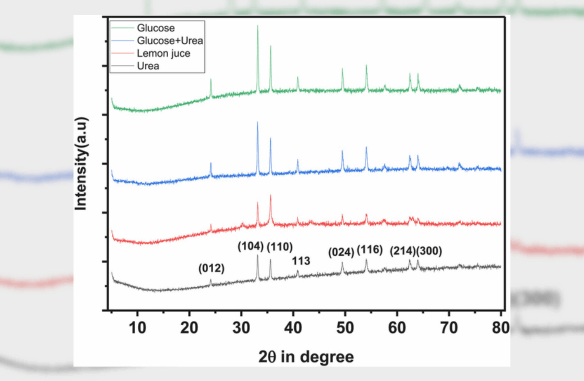
Abstract: The size- and shape-controlled synthesis of α-Fe2O3 nanoparticles was achieved via a solution combustion method using glucose, urea, and lemon juice as fuels. X-ray diffraction (XRD) confirmed the formation of α-Fe2O3 with minor γ-Fe2O3 phases, and crystallite sizes were calculated using Scherrer’s equation. SEM-EDX analysis revealed quasi-cubic morphology with uniform elemental distribution. Mössbauer spectroscopy indicated structure-dependent magnetic properties and confirmed the coexistence of α-Fe2O3 and γ-Fe2O3 phases, with evidence of superparamagnetic relaxation effects at room temperature. Density Functional Theory (DFT) calculations provided insights into the structural, electronic, and magnetic properties of bulk and surface α-Fe2O3. These findings highlight the potential of the synthesized nanoparticles for applications in magnetic devices and energy storage systems.
Author(s): V Jagadeesha Angadi, Stass Kubrin, MA Sayed, Ashok Kumar, Kuldeep Sharma, Mohd Shkir, SO Manjunatha, Renan AP Ribeiro, Marisa C Oliveira, Elson Longo, Nagaraj Basavegowda
First published: 01/09/2025