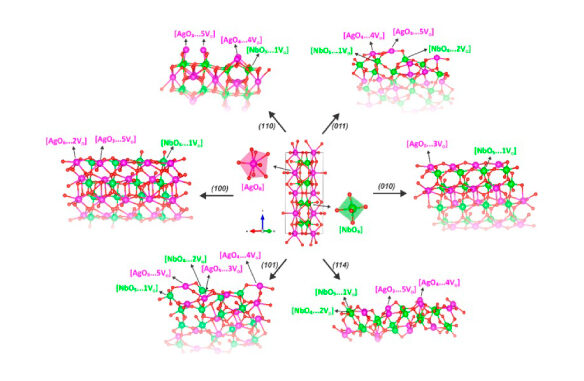
Abstract: In this work, DFT calculations have been employed to delve into the structural, electronic, and optical properties of low-index (010), (100), (101), (110), (011), and (114) surfaces of AgNbO3. Wulff construction was used to predict the available morphologies of this material and their transformations, which were matched with the experimental images obtained by electron microscopy to support our findings. Our data indicate that the undercoordinated O anions and Ag and Nb cations on these surfaces act as frustrated Lewis base and acid pairs, respectively, to control their structure and electronic properties. These sites at the (110) and (010) selectively bind H2O and O2 molecules, opening an energetically favorable pathway for the dissociation of H2O to enhance the initial stages of the formation of reactive oxygen species, ⋅OH, ⋅O2− and ⋅OOH radicals, which adsorbed strongly on both surfaces within a simplified model. Overall, the results demonstrate that careful consideration of the impacts of surface chemistry on the behavior of AgNbO3 surfaces is required to further understand and tailor the reactivity based on the generation of these highly reactive species.
Author(s): Marisa Carvalho de Oliveira, Elson Longo, Renan A.P. Ribeiro, Samantha C.S. Lemos, Juan Andrés, Lourdes Gracia
Computational Materials Science
First published: 1/1/2025
https://doi.org/10.1016/j.commatsci.2024.113398
CDMF
The CDMF, hosted at the Federal University of São Carlos (UFSCar), is one of the Research, Innovation and Dissemination Centers (RIDC) supported by the São Paulo State Research Support Foundation (Fapesp), and also receives investment from the National Council Scientific and Technological Development (CNPq), from the National Institute of Science and Technology of Materials in Nanotechnology (INCTMN).