Writers: Paulo A. Raymundo-Pereira, Marcos F.S. Teixeira, Orlando Fatibello-Filho, Edward R. Dockal, Viviane Gomes Bonifácio, Luiz H. Marcolino-Junior
Keywords: Ranitidine; Modified carbon paste electrode; Oxovanadium (IV) complex of salen; Electrochemical sensor
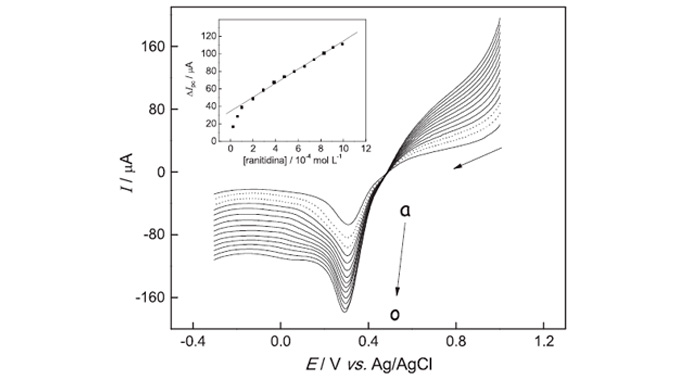
Abstract: The preparation and electrochemical characterization of a carbon paste electrode modified with the N,N-ethylene-bis(salicyllideneiminato)oxovanadium (IV) complex ([VO(salen)]) as well as its application for ranitidine determination are described. The electrochemical behavior of the modified electrode for the electroreduction of ranitidine was investigated using cyclic voltammetry, and analytical curves were obtained for ranitidine using linear sweep voltammetry (LSV) under optimized conditions. The best voltammetric response was obtained for an electrode composition of 20% (m/m) [VO(salen)] in the paste, 0.10 mol L− 1 of KCl solution (pH 5.5 adjusted with HCl) as supporting electrolyte and scan rate of 25 mV s− 1. A sensitive linear voltammetric response for ranitidine was obtained in the concentration range from 9.9 × 10− 5 to 1.0 × 10− 3 mol L− 1, with a detection limit of 6.6 × 10− 5 mol L− 1 using linear sweep voltammetry. These results demonstrated the viability of this modified electrode as a sensor for determination, quality control and routine analysis of ranitidine in pharmaceutical formulations.